Ga-Gepatonin in Brief
GA-Gepatonin now pass registration at the Pharmacological Committee of the Ministry of Health and Social Affairs of Georgia.
GA-Gepatonin has passed preclinical and clinical trials in following organizations:
GA-Gepatonin is recommended for the treatment of cirrhosis, hepatitis-A,B,C and over liver disorders.
Clinical trials show that use of the GA- Gepatonin in the treatment of patients with chronic hepatitis C possesses rather high efficiency in treatment of patients with virus hepatitis C disease:
Molecular Mechanism of the GA- Gepatonin Anti-Hepatitis C Action.
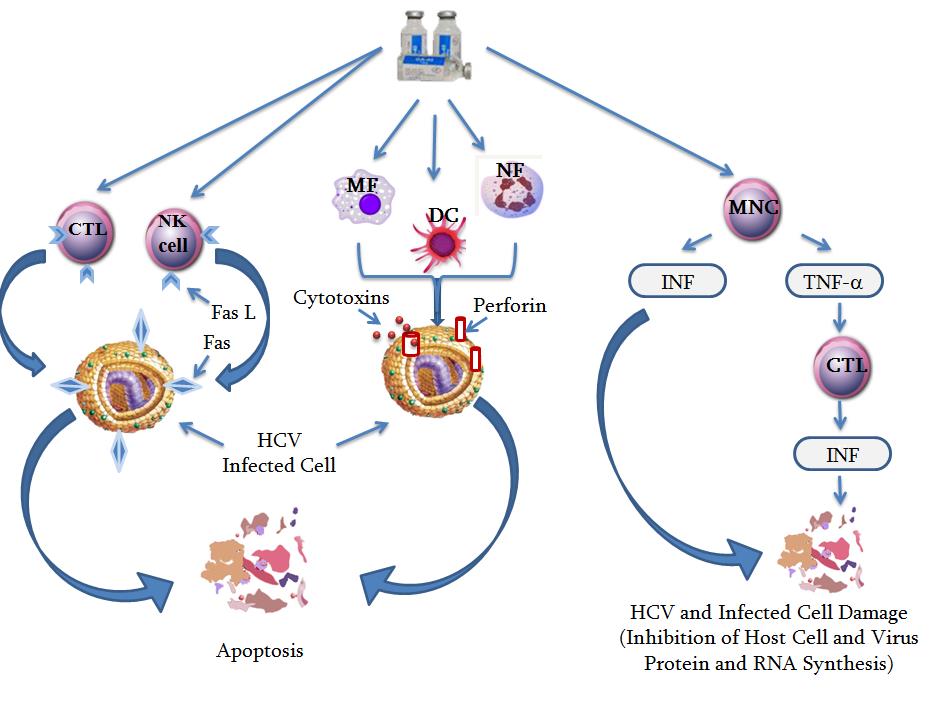
Getting (penetration) into the body hepatitis C virus causes suppression of the immune system by inhibiting Interferon’s (IFN) production by mononuclear immune cells. Interferon’s inhibit protein synthesis within the infected cells and reduce viral replication in host cells. Inhibition protein synthesis destroys both the virus and infected host cells and restricts the entry of the virus in neighboring cells. Another function of interferon’s, such as interferon-γ (INF-γ), is to directly activate mononuclear immune cells, such as T-killer, NK-cells, macrophages and others.
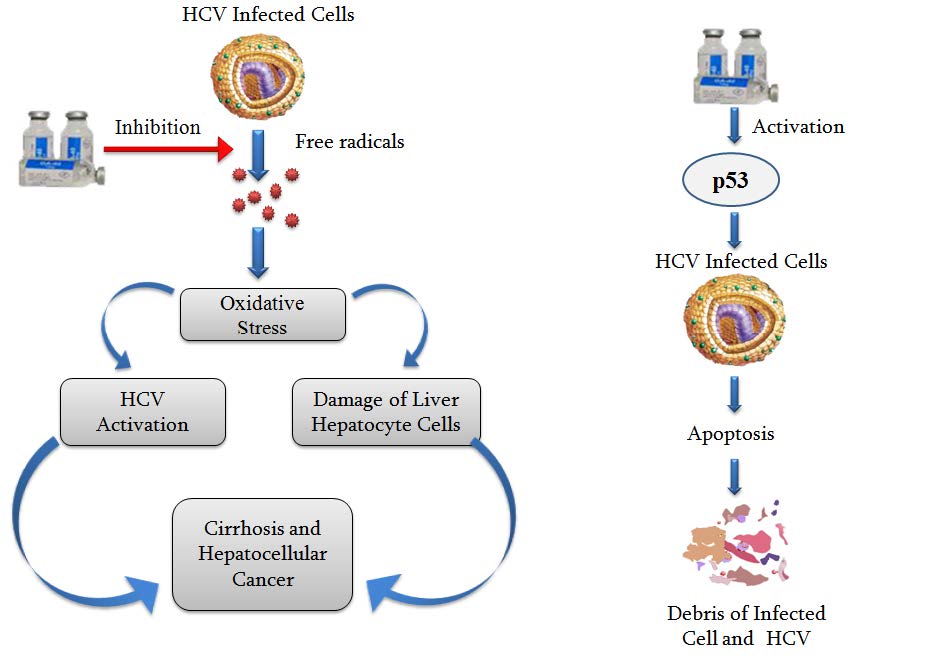
Activated cells release cell lytic cytotoxins perforin, granzymes, and granulysin and trigger apoptosis, programmed cell death of the hepatitis C virus infected target cells. Viral spread also limit by increasing p53 activity, which kills virus-infected cells by promoting apoptosis. Release of IFN-ƴ from cells is also induced by cytokines, such as interleukin 1, interleukin 2, interleukin 12; tumor necrosis factor can also enhance interferon-ƴ production. During prolonged HCV infection many complex processes play critical roles in the development of liver damage. One of these processes includes enhanced production of free radicals, causing liver oxidative stress, increase HCV disease activity in chronic hepatitis, causes liver injury leading to fibrosis and eventually hepatocellular carcinoma.
The results of pre and clinical trials of the medical preparation GA-Gepatonin, allows to submit a scheme of the molecular mechanisms GA-Gepatonin anti-hepatitis C virus-action, which leads to the high efficiency of the GA-Gepatonin treatment of patients with chronic hepatitis C virus disease.
In the treatment of chronic hepatitis C using GA-Gepatonin crossing an inactive form of the disease occurs from 1 to 7 treatments for all patients. In addition, the transition to an inactive form has a permanent effect. In patients treated with the control exerted over four years and in 95% of cases (for lack of provoking factor) activation was not found.
After four courses of treatment recommended control analysis of the presence of virus (PCR). When negative results every six months during the two years it is necessary to operate the control PCR.
If after four treatments PCR showed the presence of virus, hepatitis should be continued, and after seven treatments once again make PCR analysis.
The end result of the total elimination of the virus occurs in 60% of cases.
The amount of therapy depends on the severity of the person and the dynamics of treatment.
Cirrhosis
When using the preparation GA-Gepatonin in most cases achieved regression of disease as evidenced by the fact that the preparation GA-Gepatonin has very strong hepatoprotektorna characteristics and affects the recovery process of the exchange of substances (protein, lipid,
and carbohydrate). Regardless of the etiology of cirrhosis (viral, alcoholic, autoimmune) improve the condition occurs in 91% of cases.
The amount of therapy depends on the severity of the person and the dynamics of treatment.
GA-Gepatonin has passed preclinical and clinical trials in following organizations:
- The Kiev Medical Institute named after Bogomolts, Department Infectious Diseases, Kiev, Ukraine.
- Medical Center Diseases "Dobrobutt", Department Infectious Diseases, Kiev, Ukraine.
- Clinical and Experimental Research Institute of Tbilisi State Medical University;
- Medical & Biological Scientific Research Centre – Company <<Alexis>>;
- Laboratory of Cell Signal Mechanisms of the I.Paladin Institute of Biochemistry at Ukrainian Medical Academy;
- Medical Research Institute of Physical Chemistry at the Ministry of Healthcare of Russian Federation;
- Pharmacological Department of the Medical University of Russian Federation;
- Fuji Memorial Institute of the Preclinical Research, Bivako/Japan;
- Tokushima Otsuka Immunology Research Institute, Japan;
- Tokushima Institute of New Drug Research and Safety Evaluation, Japan;
GA-Gepatonin is recommended for the treatment of cirrhosis, hepatitis-A,B,C and over liver disorders.
Clinical trials show that use of the GA- Gepatonin in the treatment of patients with chronic hepatitis C possesses rather high efficiency in treatment of patients with virus hepatitis C disease:
- Unpleasant feelings in the right hypochondrium disappeared, the general condition considerably improved.
- Expressed positive dynamics of indicators of ALT, AST, thymol test, GGT, the general bilirubin is obvious.
- Application of GA- Gepatonin is expedient at patients with chronic hepatitis C which have contraindications to carrying out PVT.
- GA- Gepatonin action does not depend on a genotype of a virus of hepatitis C, but to some extent depends on a stage of phibrosis of liver: clinical and biochemical improvement was more expressed at phibrosis F2 and F3.
- Most likely GA- Gepatonin can be applied to radical treatment of chronic hepatitis C , practically without contraindications. It is not excluded that GA- Gepatonin in a combination with PVT can be applied in treatment not only for not respondents, but also persons with an adverse genotype of IL28 v, at which efficiency of standard PVT within 20-25%.
- In 2 months after completion of treatment the PCR-test for HCV remained negative with 60% of patients.
Molecular Mechanism of the GA- Gepatonin Anti-Hepatitis C Action.
Getting (penetration) into the body hepatitis C virus causes suppression of the immune system by inhibiting Interferon’s (IFN) production by mononuclear immune cells. Interferon’s inhibit protein synthesis within the infected cells and reduce viral replication in host cells. Inhibition protein synthesis destroys both the virus and infected host cells and restricts the entry of the virus in neighboring cells. Another function of interferon’s, such as interferon-γ (INF-γ), is to directly activate mononuclear immune cells, such as T-killer, NK-cells, macrophages and others.
Activated cells release cell lytic cytotoxins perforin, granzymes, and granulysin and trigger apoptosis, programmed cell death of the hepatitis C virus infected target cells. Viral spread also limit by increasing p53 activity, which kills virus-infected cells by promoting apoptosis. Release of IFN-ƴ from cells is also induced by cytokines, such as interleukin 1, interleukin 2, interleukin 12; tumor necrosis factor can also enhance interferon-ƴ production. During prolonged HCV infection many complex processes play critical roles in the development of liver damage. One of these processes includes enhanced production of free radicals, causing liver oxidative stress, increase HCV disease activity in chronic hepatitis, causes liver injury leading to fibrosis and eventually hepatocellular carcinoma.
The results of pre and clinical trials of the medical preparation GA-Gepatonin, allows to submit a scheme of the molecular mechanisms GA-Gepatonin anti-hepatitis C virus-action, which leads to the high efficiency of the GA-Gepatonin treatment of patients with chronic hepatitis C virus disease.
In the treatment of chronic hepatitis C using GA-Gepatonin crossing an inactive form of the disease occurs from 1 to 7 treatments for all patients. In addition, the transition to an inactive form has a permanent effect. In patients treated with the control exerted over four years and in 95% of cases (for lack of provoking factor) activation was not found.
After four courses of treatment recommended control analysis of the presence of virus (PCR). When negative results every six months during the two years it is necessary to operate the control PCR.
If after four treatments PCR showed the presence of virus, hepatitis should be continued, and after seven treatments once again make PCR analysis.
The end result of the total elimination of the virus occurs in 60% of cases.
The amount of therapy depends on the severity of the person and the dynamics of treatment.
Cirrhosis
When using the preparation GA-Gepatonin in most cases achieved regression of disease as evidenced by the fact that the preparation GA-Gepatonin has very strong hepatoprotektorna characteristics and affects the recovery process of the exchange of substances (protein, lipid,
and carbohydrate). Regardless of the etiology of cirrhosis (viral, alcoholic, autoimmune) improve the condition occurs in 91% of cases.
The amount of therapy depends on the severity of the person and the dynamics of treatment.
CTL- T-killer cells , NK-cell- natural killer cell, MF- macrophage, DK-dendrites’ cell, NF-neutrophils, Cytotoxin-granzymes, and granulysin, Fas L - is a homotrimeric type II CTL an NK-cell transmembrane protein. Fas L recognize, interact and bind with Fas (death receptor), which spans the membrane of the cancer or infected "target" cells. This high specifically interaction between Fas L and Fas surface receptors usually leads to apoptosis, or cell death.
GA- Gepatonin activates mononuclear cells, cytotoxic-T cells (T-killer cell) s, T-helper, Natural killer cells (NK-cells), macrophages, neutrophils. Activated cells release infected cell lytic perforin and cytotoxin-granzymes, and granulysin and trigger apoptosis - programmed cell death of the hepatitis C virus infected target cells. A second way to induce apoptosis is via cell-surface interactions between the T-killer cells, NK-cells cells and the infected cells.
The final result is apoptosis of the hepatitis C virus infected target cells.
GA- Gepatonin activates mononuclear cells, cytotoxic-T cells (T-killer cell) s, T-helper, Natural killer cells (NK-cells), macrophages, neutrophils. Activated cells release infected cell lytic perforin and cytotoxin-granzymes, and granulysin and trigger apoptosis - programmed cell death of the hepatitis C virus infected target cells. A second way to induce apoptosis is via cell-surface interactions between the T-killer cells, NK-cells cells and the infected cells.
The final result is apoptosis of the hepatitis C virus infected target cells.
- GA-Gepatonin increase p53 activity, which kills virus-infected cells by promoting apoptosis of the hepatitis C virus infected target cells.
- GA- Gepatonin activates production of Tumor Necrosis Factor (TNF-α), which induce release Interferon-ƴ. Both cytokines TNF-α and IFN-ƴ kills virus-infected cells by promoting apoptosis of the hepatitis C virus infected target cells.
- GA- Gepatonin with high Anti-oxidant activity neutralizes free radicals and reduces the oxygen with two unpaired electrons, inhibit oxygen stress and decrease chronic HCV disease activity in patients.
Download Pdf Version |
| ||||||
Download Microsoft Word Version |
| ||||||